The nutraceutical industry bridges the gap between food and medicine, offering consumers natural health products. However, ensuring the quality and safety of these products relies heavily on robust document traceability throughout the supply chain. This blog explores the importance of document traceability in the nutraceutical industry, the challenges associated with it, and how Asterdocs,, empowers businesses to achieve efficient and transparent document tracking.
Why Traceability Matters in Nutraceuticals: Building Consumer Trust
Document traceability refers to the ability to track the origin, movement, and transformation of a product and its ingredients throughout the supply chain. Here’s why traceability is crucial for nutraceutical companies:
-
-
Consumer Safety & Quality Assurance: Traceability allows for the identification and location of specific batches of ingredients in case of recalls or quality issues, ensuring consumer safety.
-
-
-
Regulatory Compliance: Many regulatory bodies, like the FDA, require detailed documentation of ingredient sourcing, processing, and testing. Traceability facilitates compliance with these regulations.
-
-
-
Transparency & Trust: Consumers increasingly seek transparency about the origin and quality of the ingredients in nutraceutical products. Traceability builds trust by demonstrating a commitment to ethical sourcing and responsible manufacturing practices.
-
-
-
Improved Supply Chain Efficiency: Traceability allows for faster identification of potential issues within the supply chain, enabling proactive problem-solving and improved efficiency.
-
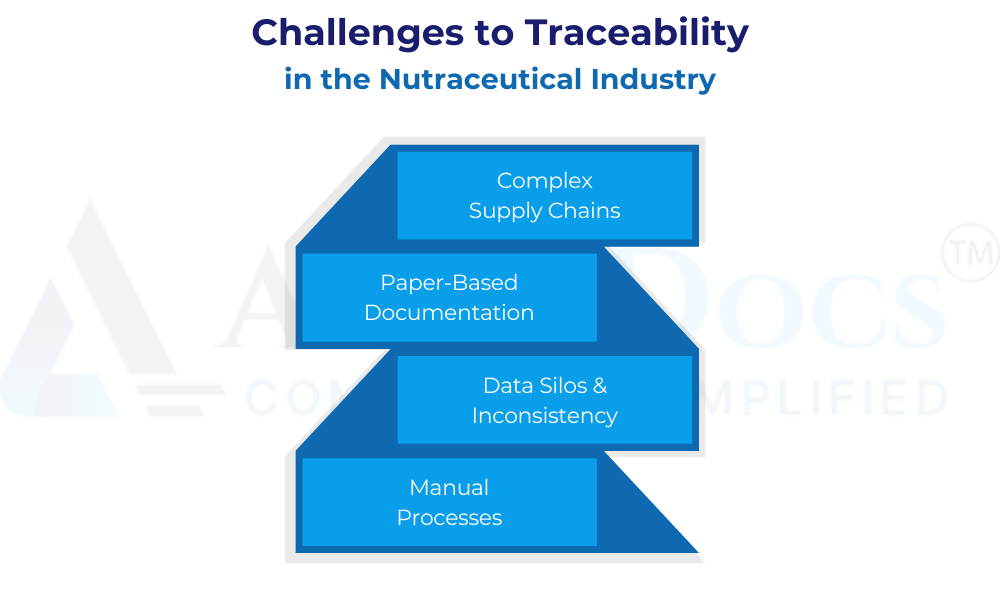
Challenges to Traceability in the Nutraceutical Industry
Despite its importance, achieving document traceability in the nutraceutical industry can be challenging:
-
- Complex Supply Chains: Global sourcing and multi-step processing can make it difficult to track the complete journey of ingredients.
-
-
Paper-Based Documentation: Reliance on paper documents can hinder easy access, retrieval, and analysis of traceability data.
-
-
-
Data Silos & Inconsistency: Scattered documentation across departments and suppliers can lead to data inconsistencies and difficulties in compiling a complete traceability record.
-
-
-
Manual Processes: Manual data entry for tracking documents can be error-prone and time-consuming.
-
-
-
Empowering Traceability: How Asterdocs Streamlines Nutraceutical Document Management
-
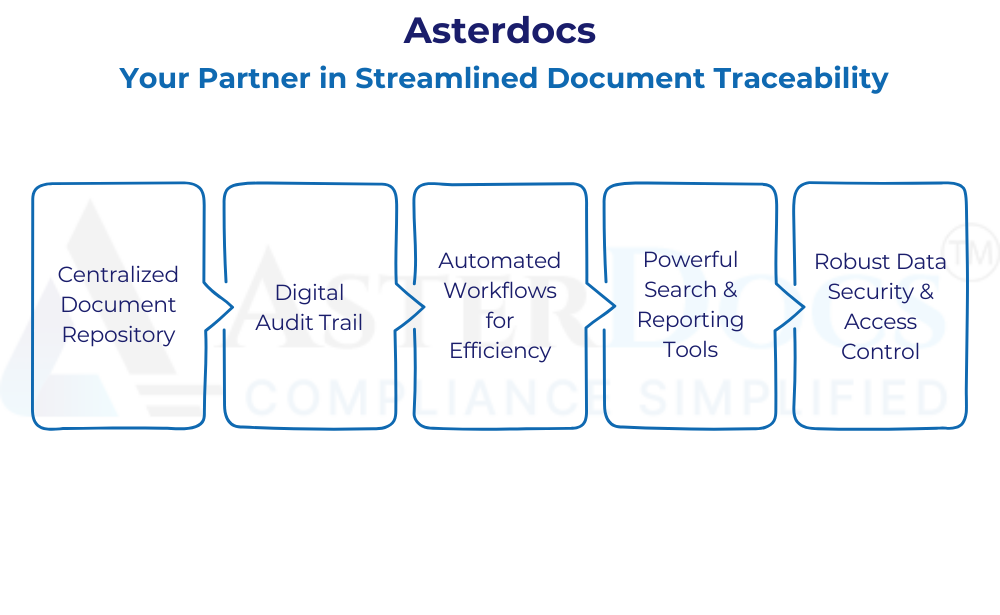
Asterdocs designed to address the specific needs of document traceability in the nutraceutical industry. Here’s how Asterdocs empowers businesses to achieve this:
-
-
Centralized Document Repository: Securely store all nutraceutical-related documents, from ingredient sourcing certificates to manufacturing batch records and quality control test results, in a single, centralized location. This facilitates easy access and retrieval for authorized personnel, streamlining traceability investigations and audits.
-
-
-
Digital Audit Trail: Maintain a clear digital audit trail for every document within Asterdocs. This allows you to track the movement of ingredients and finished products throughout the entire supply chain, from origin to final sale. This transparency is crucial for demonstrating compliance and ensuring consumer safety.
-
-
-
Automated Workflows for Efficiency: Automate repetitive tasks associated with document traceability, such as routing documents for approvals or sending notifications regarding expiring certificates. This frees up valuable employee time for more strategic tasks and minimizes the risk of human error in manual data entry.
-
-
-
Powerful Search & Reporting Tools: Asterdocs equips you with advanced search functionalities to quickly locate specific documents based on criteria like date, ingredient, supplier, or batch number. This simplifies traceability audits, streamlines investigations into potential issues, and empowers data-driven decision-making.
-
-
-
Robust Data Security & Access Control: Asterdocs prioritizes data security and integrity. Your sensitive documents are protected with robust encryption and user access controls. Permissions can be assigned based on roles and responsibilities, ensuring only authorized personnel can access specific information.
-
Conclusion: Building a Transparent and Traceable Supply Chain
By embracing Asterdocs, nutraceutical companies can establish a robust document traceability system and achieve the following benefits:
-
-
Enhanced consumer trust and brand reputation.
-
-
-
Improved efficiency and compliance with regulations.
-
-
-
Faster response times in case of recalls or quality issues.
-
-
-
Data-driven decision-making for optimizing the supply chain.
-
In today’s competitive landscape, transparency and traceability are no longer optional but essential for success in the nutraceutical industry. Asterdocs empowers businesses to build a traceable supply chain, fostering consumer trust and ensuring the highest quality products for a healthier tomorrow.